科学素养与现象阐释·英语30篇(6)
23 / 30
正在校验访问权限...
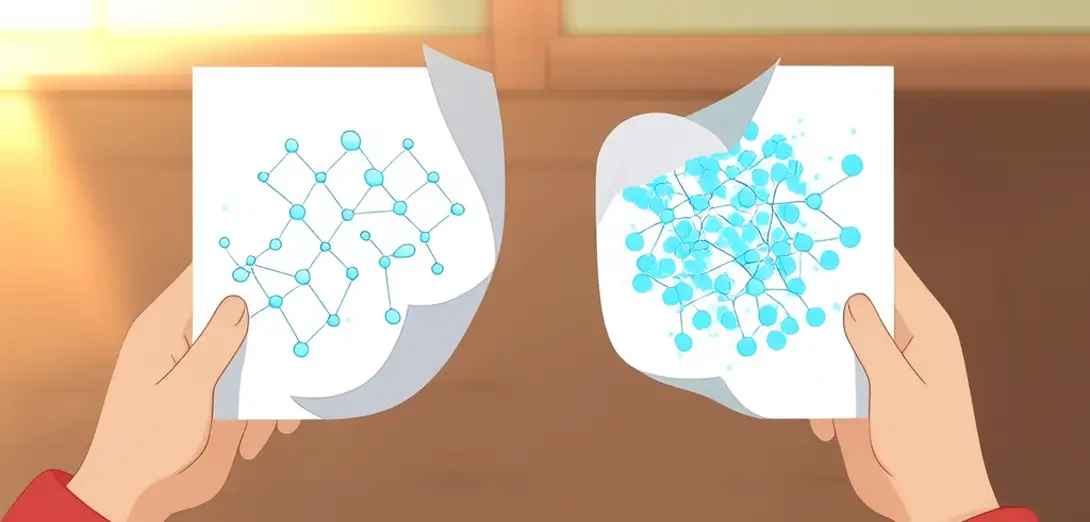
Why Wet Paper Loses Structural Integrity: Hydrogen Bond Disruption Dynamics
为什么纸张浸水后强度显著下降:氢键断裂动力学
-
Paper’s dry strength derives primarily from hydrogen bonding between cellulose microfibrils, not mechanical interlocking or lignin adhesion.
-
Water molecules infiltrate inter-fibrillar spaces, competing with and displacing existing hydrogen bonds through superior dipole interactions with hydroxyl groups.
-
Capillary action drives rapid penetration along fibril surfaces, especially in papers with high porosity and low sizing agent content.
-
Wet strength additives like polyamide-epichlorohydrin resins create covalent crosslinks resistant to hydrolysis, but these constitute <5% of total bond density.
-
Fiber swelling increases interfibrillar distances, reducing van der Waals forces and disrupting the percolating hydrogen-bond network essential for load distribution.
-
Recycled fibers exhibit greater wet weakening because repeated drying cycles permanently reduce accessible hydroxyl sites for re-bonding.
-
Relative humidity above 80% initiates measurable strength loss even without full immersion, revealing the hygroscopic sensitivity of cellulose hydration shells.
-
Industrial papermaking controls wet-web integrity via press nips and steam-heated dryers that manage water removal kinetics to preserve transient bond formation.
-
Atomic force microscopy shows hydrogen bond lifetime drops from ~10⁻⁹ seconds in dry state to ~10⁻¹² seconds in saturated cellulose.
-
This nanoscale bond instability explains macroscopic phenomena like ink bleeding and dimensional instability during printing.
-
Ultimately, paper behaves as a metastable colloidal assembly whose integrity collapses when hydration thermodynamics override intermolecular binding energies.
-
Understanding this informs sustainable packaging design where controlled moisture resistance must coexist with biodegradability requirements.